Abstract
Research Article
Antiviral activity of Eucalyptus camaldulensis leaves ethanolic extract on herpes viruses infection
Mahmoud Huleihel* and Aya Abu-Jafar
Published: 13 October, 2017 | Volume 1 - Issue 1 | Pages: 001-009
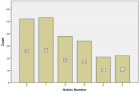
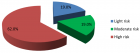
Eucalyptus camaldulensis (Ec) is considered as a traditional medicinal plant with valuable therapeutic effects. Here we evaluated the antiviral activity of its ethanolic leave extract on different herpes viruses. Vero cells were infected with either of the tested viruses [herpes simplex virus -1 and 2 (HSV-1, HSV-2) and Varicella-Zoster Virus (VZV)] with or without treatment with Ec extract and viral infection development was evaluated by plaque assay. Our results showed significant antiviral activity of the examined extract against all tested viruses. The 80%-MeOH fraction of this extract offered the highest activity against these viruses with 50% inhibitory concentration (IC50) of 0.1±0.08, 0.3±0.02 and 1±0.03 μg/ml against HSV-1, HSV-2 and VZV respectively and 50% cytotoxicity (CC50) at 700 μg/ml. The highest antiviral effect of this fraction was obtained mainly when it was added during and post infection (p.i.) or when it was added only p.i. Also, this fraction significantly reduced the amount of infective endogenous viral particles in cells that were treated with the 80%-MeOH fraction only post viral entry into the host cells. A synergistic antiviral effect against all tested viruses was also observed when cells were treated with a combination of acyclovir (ACV) and 80%-MeOH fraction of Ec. Further study is required for the isolation and identification of the anti-virally active component/s of this fraction.
Read Full Article HTML DOI: 10.29328/journal.ijcv.1001001 Cite this Article Read Full Article PDF
Keywords:
Eucalyptus camaldulensis; Ethanolic extract; HSV; VZV; Acyclovir
References
- Müller V, Chávez JH, Reginatto FH, Zucolotto SM, Niero R, et al. Evaluation of antiviral activity of South American plant extracts against herpes simplex virus type 1 and rabies virus. Phytother Res. 2007; 10: 970-974. Ref: https://goo.gl/Y5BY1V
- Elion GB, Furman PA, Fyfe JA, de Miranda P, Beauchamp L, et al. Selectivity of action of an antiherpetic agent, 9-(2-hydroxyethoxymethyl)guanine. Proc Natl Acad Sci U S A. 1977; 12: 5716-5720. Ref: https://goo.gl/hFkJCr
- Meyers JD, Wade JC, Mitchell CD, Saral R, Lietman PS, et al. Multicenter collaborative trial of intravenous acyclovir for treatment of mucocutaneous herpes simplex virus infection in the immunocompromised host. Am J Med. 1982; 73: 229-235. Ref: https://goo.gl/meF9HM
- Snoeck R. Antiviral therapy of herpes simplex. Int J Antimicrob Agents. 2000; 16: 157-159. Ref: https://goo.gl/jhNJgQ
- Khan MT, Atherb A, Thompson KD, Gambari R. Extracts and molecules from medicinal plants against herpes simplex viruses. Antiviral Res. 2005; 67: 107-119. Ref: https://goo.gl/37YbHC
- Whitley RJ, Bernard Roizman. Herpes simplex virus infections. Lancet. 2001; 357: 1513-1518. Ref: https://goo.gl/eXTLqC
- Celum C1, Wald A, Hughes J, Sanchez J, Reid S, et al. Effect of aciclovir on HIV-1 acquisition in herpes simplex virus 2 seropositive women and men who have sex with men: a randomised, double-blind, placebo-controlled trial. Lancet. 2008; 371: 2109-2119. Ref: https://goo.gl/jeCvWE
- De Clercq E. Antiviral drugs in current clinical use. J Clin Virol. 2004; 30: 115-133. Ref: https://goo.gl/TCxt1x
- Bao B1, Meng Z, Li N, Meng Z, Zhang L, et al. Design Synthesis and Antiviral Activity Studies of Schizonepetin Derivatives. Int J Mol Sci. 2013; 14: 17193-17203. Ref: https://goo.gl/PMF8sy
- Fiddian AP, Brigden D, Yeo JM, Hickmott EA. Acyclovir: An update of the clinical applications of this antiherpes agent. Antiviral Res. 1984; 4: 99-117. Ref: https://goo.gl/utMMLp
- Erlich KS, Mills J, Chatis P, Mertz GJ, Busch DF, et al. Acyclovir-Resistant Herpes Simplex Virus Infections in Patients with the Acquired Immunodeficiency Syndrome. N Engl J Med. 1989; 320: 293-296. Ref: https://goo.gl/c7ECYP
- Oliver NM, Collins P, Van der Meer J, Van't Wout JW. Biological and biochemical characterization of clinical isolates of herpes simplex virus type 2 resistant to acyclovir. Antimicrob Agents Chemother. 1989; 33: 635-640. Ref: https://goo.gl/8XuHqe
- Burrelab S, Aimeb C, Hermetb L, Ait-Arkoub Z, Aguta H, et al. Surveillance of herpes simplex virus resistance to antivirals: A 4-year survey. Antiviral Res. 2013; 100: 365-372. Ref: https://goo.gl/nVgrTt
- Churqui MP, Lind L, Thörn K, Svensson A, Savolainen O, et al. Extracts of Equisetum giganteum L and Copaifera reticulate Ducke show strong antiviral activity against the sexually transmitted pathogen herpes simplex virus type 2. J Ethnopharmacol. 2017; 210: 192-197. Ref: https://goo.gl/Xb2H66
- Frobert E, Burrel S, Ducastelle-Lepretre S, Billaud G, Ader F, et al. Resistance of herpes simplex viruses to acyclovir: An update from a ten-year survey in France. Antiviral Res. 2014; 111: 36-41. Ref: https://goo.gl/Az5DkS
- Field AK, Biron KK. "The end of innocence" revisited: resistance of herpesviruses to antiviral drugs. Clin Microbiol Rev. 1994; 7: 1-13. Ref: https://goo.gl/VD1Hy2
- İlker Devrim, Hasan Tezer, Göknur Haliloğlu, Ateş Kara, Gülten Seçmeer. Relapsing Herpes simplex virus encephalitis despite high-dose acyclovir therapy: a case report. Turk J Pediatr. 2008; 50: 380-382. Ref: https://goo.gl/Q429CT
- Picton SF, Flatt PR, McClenaghan NH. Differential Acute and Long Term Actions of Succinic Acid Monomethyl Ester Exposure on Insulin-Secreting BRIN-BD11 Cells. Int J Exp Diabetes Res. 2001; 2: 19-27. Ref: https://goo.gl/vFehUS
- Clark A . Natural products as a resource for new drugs. Pharm Res. 1996; 13: 1133-1141. Ref: https://goo.gl/4tiws9
- Bailly C1, Perrine D, Lancelot JC, Saturnino C, Robba M, et al. Sequence-selective binding to DNA of bis(amidinophenoxy)alkanes related to propamidine and pentamidine. Biochem J. 1997; 323: 23-31. Ref: https://goo.gl/zuuDNz
- Yarmolinsky L, Zaccai M, Ben-Shabat S, Mills D, Huleihel M. Antiviral activity of ethanol extracts of Ficus binjamina and Lilium candidum in vitro. N Biotechnol. 2009; 26: 307-313. Ref: https://goo.gl/cJA2zf
- Yarmolinsky L, Huleihel M, Zaccai M, Ben-Shabat S. Potent antiviral flavone glycosides from Ficus benjamina leaves. Fitoterapia. 2012; 83: 362-367. Ref: https://goo.gl/zecCdJ
- Chiang LC, Cheng HY, Liu MC, Chiang W, Lin CC. In Vitro anti-herpes simplex viruses and anti-adenoviruses activity of twelve traditionally used medicinal plants in Taiwan. Biol Pharm Bull. 2003; 26: 1600-1604. Ref: https://goo.gl/aZGTbE
- Jassim SA, Naji MA. Novel antiviral agents: a medicinal plant perspective. J Appl Microbiol. 2003; 95: 412-427. Ref: https://goo.gl/3gLiUo
- Sanchez Palomino S, Abad MJ, Bedoya LM, García J, Gonzales E, et al. Screening of South American Plants against Human Immunodeficiency Virus: Preliminary Fractionation of Aqueous Extract from Baccharis trinervis. Biol Pharm Bull. 2002; 25: 1147-1150. Ref: https://goo.gl/QXee6r
- Elaissi A, Rouis Z, Salem NA, Mabrouk S, ben Salem Y, et al. Chemical composition of 8 eucalyptus species' essential oils and the evaluation of their antibacterial, antifungal and antiviral activities. BMC Complement Altern Med. 2012; 12: 81-81. Ref: https://goo.gl/7bTF4H
- Gemechu A, Giday M, Worku A, Ameni G. In vitro Anti-mycobacterial activity of selected medicinal plants against Mycobacterium tuberculosis and Mycobacterium bovis Strains. BMC Complement Altern Med. 2013; 13: 291-291. Ref: https://goo.gl/kcmWeU
- Almeida IF, Fernandes E, Lima JL, Valentão P, Andrade PB, et al. Oxygen and Nitrogen Reactive Species Are Effectively Scavenged by Eucalyptus globulus Leaf Water Extract. J Med Food. 2009; 12: 175-183. Ref: https://goo.gl/S8AWVa
- Takasaki M, Konoshima T, Fujitani K, Yoshida S, Nishimura H, et al. Inhibitors of skin-tumor promotion. VIII. Inhibitory effects of euglobals and their related compounds on Epstein-Barr virus activation. (1). Chem Pharm Bull (Tokyo). 1990; 38: 2737-2739. Ref: https://goo.gl/xeVpFM
- Brochot A, Guilbot A, Haddioui L, Roques C. Antibacterial, antifungal, and antiviral effects of three essential oil blends. Microbiologyopen. 2017; 6: e00459. Ref: https://goo.gl/SxkZDH
- Dhifi W, Bellili S, Jazi S, Bahloul N, Mnif W. Essential Oils’ Chemical Characterization and Investigation of Some Biological Activities: A Critical Review. Medicines (Basel). 2016; 3: 25. Ref: https://goo.gl/7WX1ck
- Döll-Boscardin PM, Sartoratto A, Sales Maia BH, Padilha de Paula J, Nakashima T, et al. In Vitro Cytotoxic Potential of Essential Oils of Eucalyptus benthamii and Its Related Terpenes on Tumor Cell Lines. Evid Based Complement Altern Med. 2012; 2012: 342652. Ref: https://goo.gl/FpGa7E
- Astani A, Reichling J, Schnitzler P. Comparative Study on the Antiviral Activity of Selected Monoterpenes Derived from Essential Oils. Phytother Res. 2010; 24: 673-679. Ref: https://goo.gl/2MQqoB
- Adeniyi BA, OO Ayepola, FD Adu. The antiviral activity of leaves of Eucalyptus camaldulensis (Dehn) and Eucalyptus torelliana (R. Muell). Pak J Pharm Sci. 2015; 28: 1773-1776. Ref: https://goo.gl/haPqTW
- Ju HQ, Wang SY, Pei Y, Xiang YF, Li S, et al. [In vitro study on the anti-HSV-1 and HBV activities of extracts from the fruit of Eucalyptus maidenii]. Zhong Yao Cai. 2011; 34: 242-245. Ref: https://goo.gl/6XJw8E
- YufangShi, Barbara S.Kornovski, RashminSavani, Eva ATurley. A rapid. multiwell colorimetric assay for chemotaxis. J Immunol Methods. 1993; 164: 149-154. Ref: https://goo.gl/FJss6e
- Mahmoud Huleihel, Vladimir Ishanu, Jacov Tal, Shoshana (Malis) Arad. Antiviral effect of red microalgal polysaccharides on Herpes Simplex and Varicella zoster viruses. J Appl Phycology. 2001; 13: 127-134. Ref: https://goo.gl/MksrhV
- Kirsten Geschera, Joachim Kühnb, Wali Hafezi, bAndreas Louisa, Andrea Derksen, et al. Inhibition of viral adsorption and penetration by an aqueous extract from Rhododendron ferrugineum L. as antiviral principle against herpes simplex virus type-1. Fitoterapia. 2011; 82: 408-413. Ref: https://goo.gl/u4riYt
- Noam R, M Huleihel, Michele Zaccai. Stress conditions during plant growth increase the anti-herpetic properties of Lilium candidum leaf extracts and fractions. J Med Plants Res. 2015; 9: 954-961. Ref: https://goo.gl/4whu4n
- Cheng HY, Lin LT, Huang HH, Yang CM, Lin CC. Yin Chen HaoTang, a Chinese prescription, inhibits both herpes simplex virus type-1 and type-2 infections in vitro. Antiviral Res. 2008; 77: 14-19. Ref: https://goo.gl/kPZ5Wq
- Arena A, Bisignano G, Pavone B, Tomaino A, Bonina FP, et al. Antiviral and immunomodulatory effect of a lyophilized extract of Capparis spinosa L. buds. Phytother Res. 2008; 22: 313-317. Ref: https://goo.gl/qv7bUX
- Cheng HY, Yang CM, Lin TC, Lin LT, Chiang LC, et al. Excoecarianin, Isolated from Phyllanthus urinaria Linnea, Inhibits Herpes Simplex Virus Type 2 Infection through Inactivation of Viral Particles. Evid Based Complement Alternat Med. 2011; 2011: 259103. Ref: https://goo.gl/LXreMR
- Yang CM, Cheng HY, Lin TC, Chiang LC, Lin CC. Acetone, ethanol and methanol extracts of Phyllanthus urinaria inhibit HSV-2 infection in vitro. Antiviral Res. 2005; 67: 24-30. Ref: https://goo.gl/ixSvJK
- Wyrwicz LS, Rychlewski L. Identification of Herpes TATT-binding protein. Antiviral Res. 2007; 75: 167-172. Ref: https://goo.gl/4Qi1kA
- Field HJ, S Biswas, IT Mohammad. Herpesvirus latency and therapy--from a veterinary perspective. Antiviral Res. 2006; 71: 127-33. Ref: https://goo.gl/U3kGGg
- Kesharwani A, Polachira SK, Nair R, Agarwal A, Mishra NN, et al. Anti-HSV-2 activity of Terminalia chebula Retz extract and its constituents, chebulagic and chebulinic acids. BMC Complement Altern Med. 2017; 17: 110. Ref: https://goo.gl/UTsnZZ
- Gescher K, Kühn J, Lorentzen E, Hafezi W, Derksen A, et al. Proanthocyanidin-enriched extract from Myrothamnus flabellifolia Welw. exerts antiviral activity against herpes simplex virus type 1 by inhibition of viral adsorption and penetration. J Ethnopharmacol. 2011; 134: 468-474. Ref: https://goo.gl/Fe1Yc3
- Jabareen A, M Huleihil, M Huleihel. Effect of Extracts of Passiflora edulis Leaves on Herpes Viruses Infection. J Virol Antivir Res. 2013; 2: 1-6. Ref: https://goo.gl/JcX4jG
- Choi HJ, Song JH, Park KS, Kwon DH. Inhibitory effects of quercetin 3-rhamnoside on influenza A virus replication. Eur J Pharm Sci. 2009; 37: 329-333. Ref: https://goo.gl/1BrUuS
- Mucsi I, Prágai BM. Inhibition of virus multiplication and alteration of cyclic AMP level in cell cultures by flavonoids. Experientia. 1985; 41: 930-931. Ref: https://goo.gl/GGqxUA
Figures:
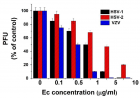
Figure 1
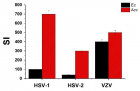
Figure 2
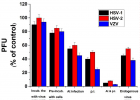
Figure 3
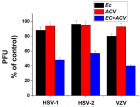
Figure 4
Similar Articles
-
Antiviral activity of Eucalyptus camaldulensis leaves ethanolic extract on herpes viruses infectionMahmoud Huleihel*,Aya Abu-Jafar. Antiviral activity of Eucalyptus camaldulensis leaves ethanolic extract on herpes viruses infection. . 2017 doi: 10.29328/journal.ijcv.1001001; 1: 001-009
Recently Viewed
-
Cervical choriocarcinoma in a post-menopause woman: Case report and review of literatureZeinab Nazari,Leila Mortazavi*,Noushin Gordani. Cervical choriocarcinoma in a post-menopause woman: Case report and review of literature. Clin J Obstet Gynecol. 2022: doi: 10.29328/journal.cjog.1001101; 5: 019-021
-
To compare serum Vitamin D status in pre-eclamptic and non-preeclamptic pregnant women in labour: A tertiary care centre study of Northern IndiaMonica Karpa,Sita Thakur,Kamal Singh*,Jyoti Sharma,Harsha Chaudhary. To compare serum Vitamin D status in pre-eclamptic and non-preeclamptic pregnant women in labour: A tertiary care centre study of Northern India. Clin J Obstet Gynecol. 2022: doi: 10.29328/journal.cjog.1001100; 5: 013-018
-
Non-surgical Treatment of Verrucous Hyperplasia on Amputation Stump: A Case Report and Literature ReviewSajeda Alnabelsi*, Reem Hasan, Hussein Abdallah, Suzan Qattini. Non-surgical Treatment of Verrucous Hyperplasia on Amputation Stump: A Case Report and Literature Review. Ann Dermatol Res. 2024: doi: 10.29328/journal.adr.1001034; 8: 015-017
-
Outpatient operative hysteroscopy: evaluation of patient satisfaction and acceptanceClare Margaret Crowley*,Noelle Gill,Minna Geisler. Outpatient operative hysteroscopy: evaluation of patient satisfaction and acceptance. Clin J Obstet Gynecol. 2022: doi: 10.29328/journal.cjog.1001098; 5: 005-008
-
Predictors of positive treatment response to PTNS in women with overactive bladderSuneetha Rachaneni*,Doyo Enki,Megan Welstand,Thomasin Heggie,Anupreet Dua. Predictors of positive treatment response to PTNS in women with overactive bladder. Clin J Obstet Gynecol. 2022: doi: 10.29328/journal.cjog.1001097; 5: 001-004
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."